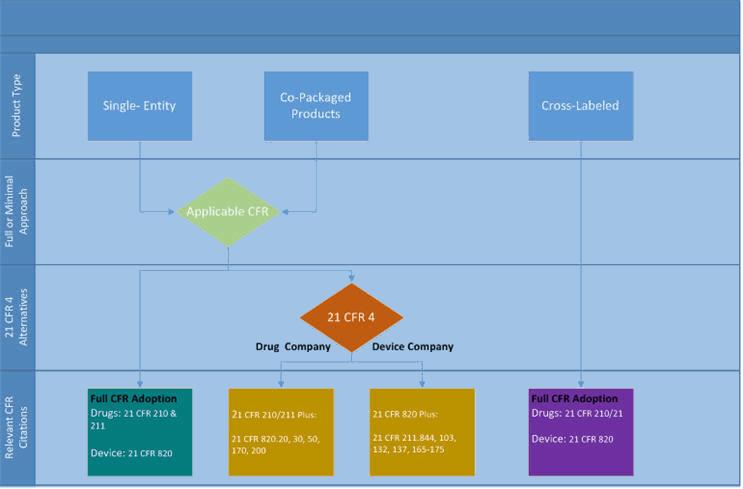
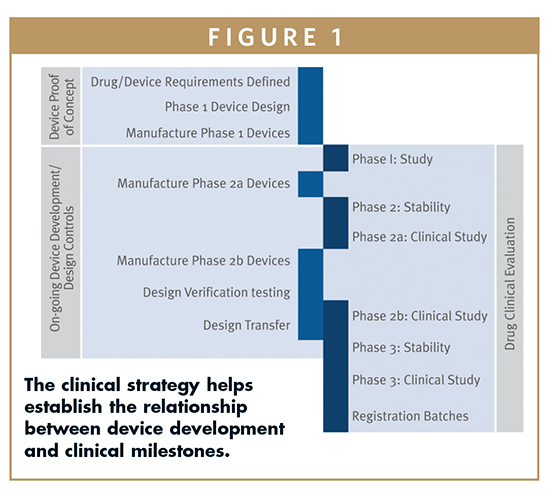
Product Development and Manufacturing Challenges for Combination Products | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

PPT - Combination Products: Cross Labeling and Single Entity Labeling PowerPoint Presentation - ID:726338

PPT - Combination Products: Cross Labeling and Single Entity Labeling PowerPoint Presentation - ID:726338

Notice: Human Drugs: Drug Products Withdrawn From Sale For Reasons Other Than Safety or Effectiveness— Combination Products and Mutually Conforming Labeling Cross Labeling | PDF | Institutional Review Board | Federal Food

1 Cross Labeling Combination Products and User Fees Bradley Merrill Thompson, MBA, JD, RAC Epstein Becker & Green PC Pharma MedDevice 2007 Conference New. - ppt download
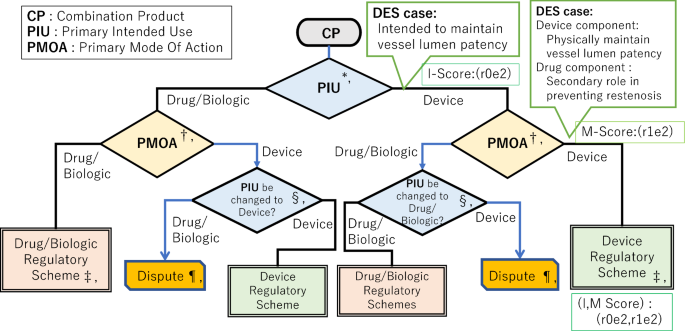
New Visualization Models of Designation Pathway and Group Categorization of Device–Drug and Device–Biologic Combination Products Classification in the United States: Analysis of FDA Capsular Decisions | SpringerLink