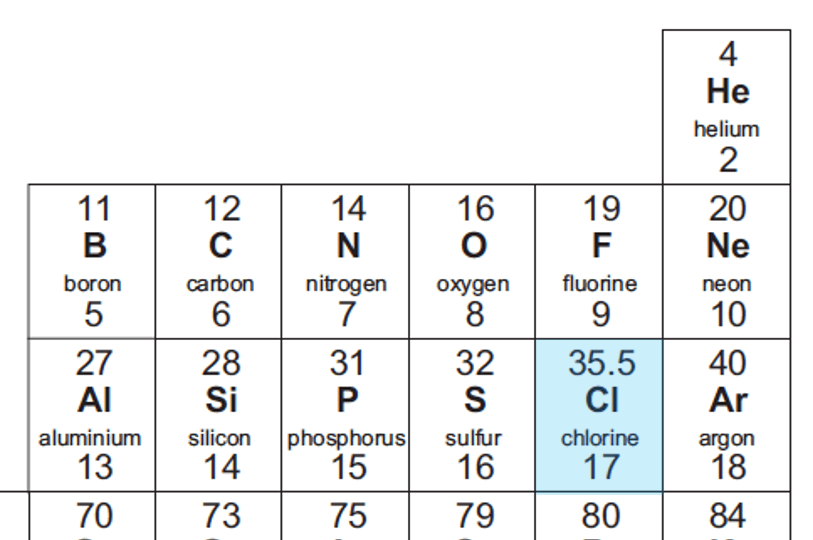
Chlorine has isotopes [math]Cl^{35}[/math] and [math]Cl^{37}[/math]. There are three [math]Cl^{35}[/math] isotopes for every one Cl isotope in a sample of chloride. What is the atomic mass of chlorine? - Quora

Defining how to calculate relative atomic mass of element relative isotopic mass definition gcse chemistry Calculations igcse O Level revision notes

Chlorine And Ph Meter Drinking Water Ram Chipset Digital Ph Meter For Aquarium - Buy Ph Meter Accessories,Ph Meter Water Proof,Smart Sensor Ph Meter Product on Alibaba.com
Chlorine exists mainly as two isotopes, 37Cl and 35Cl. Which is more abundant? How do you know? - Quora