SciELO - Saúde Pública - Readability of medicinal package leaflets: a systematic review Readability of medicinal package leaflets: a systematic review

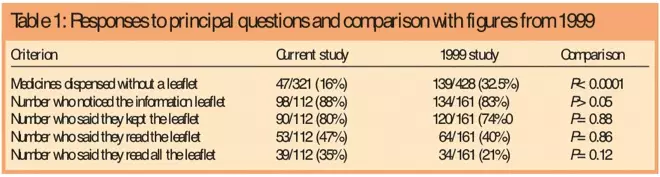
PDF) Testing of the readability of package leaflets as an initial step under the pharmaceutical care initiative towards increasing the safety of medicinal products

Why patient information leaflets have their place in informing patients about their medicines - The Pharmaceutical Journal
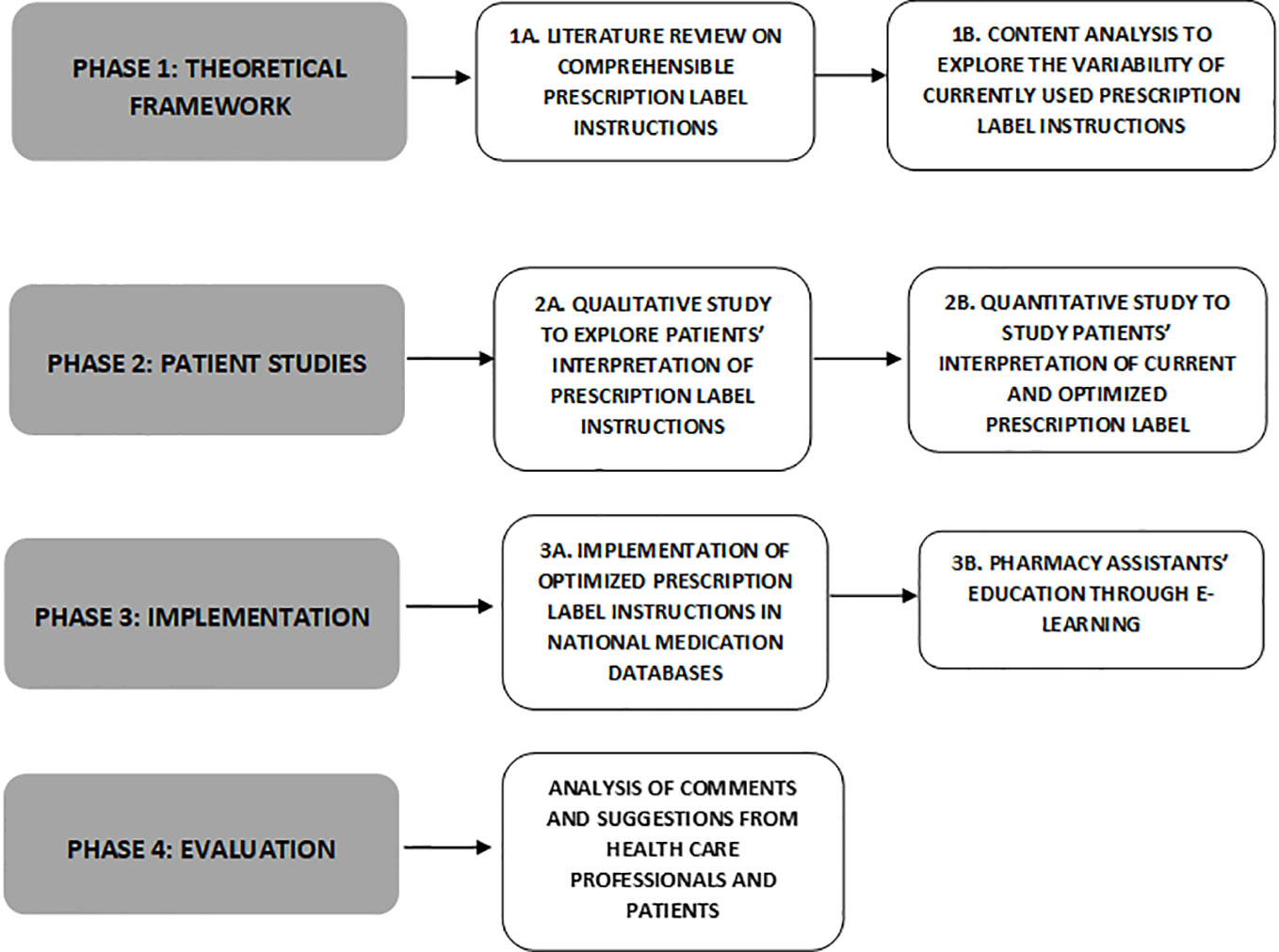
Frontiers | Development of Comprehensible Prescription Label Instructions: A Study Protocol for a Mixed-Methods Approach | Pharmacology

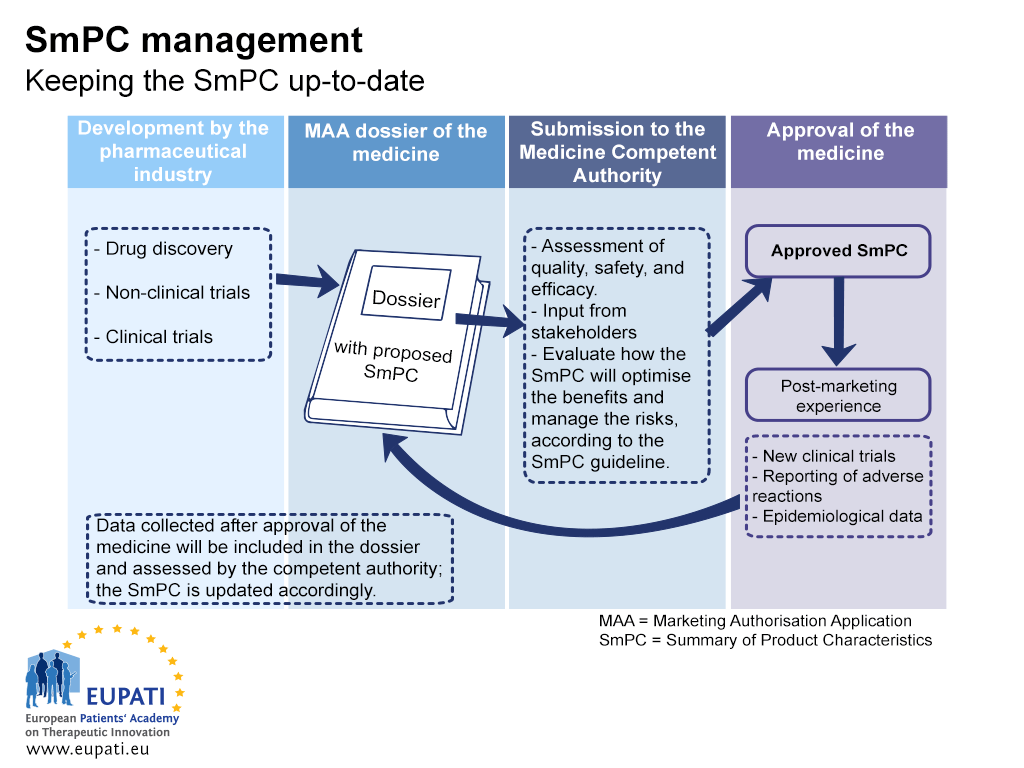
GUIDELINE ON THE READABILITY OF THE LABELLING AND LAYOUT OF THE INFORMATION ... 3.1 User testing ... The

PDF) Testing of the readability of package leaflets as an initial step under the pharmaceutical care initiative towards increasing the safety of medicinal products